|
Calcified hard tissues (e.g. bone, cartilage, eggshells) represent a key innovation in the evolution of vertebrates, playing a major part in their locomotion, physiology, and reproduction. Since their microstructure can be preserved for hundreds of millions of years, these tissues often represent the only available material to investigate extinct vertebrate biodiversity, and can give use insight into the ontogenetic, metabolic, and biomechanical constraints that influence their phenotypic variation.
I study microstructural traits in hard tissues of extant and extinct vertebrates using histology, microscopy, and X-ray computed tomography techniques, and reconstruct their evolution in a phylogenetic context with cutting-edge quantitative approaches. Integrating these variables in a multivariate framework allows me to infer their respective roles in major transitions in vertebrate evolution.
|
Below is a description of some of my current and past research projects. For a whole bibliography of my papers, see Publications.
Evolution of eggshell microstructure in reptiles, with a focus on dinosaurs including birds (since 2018)
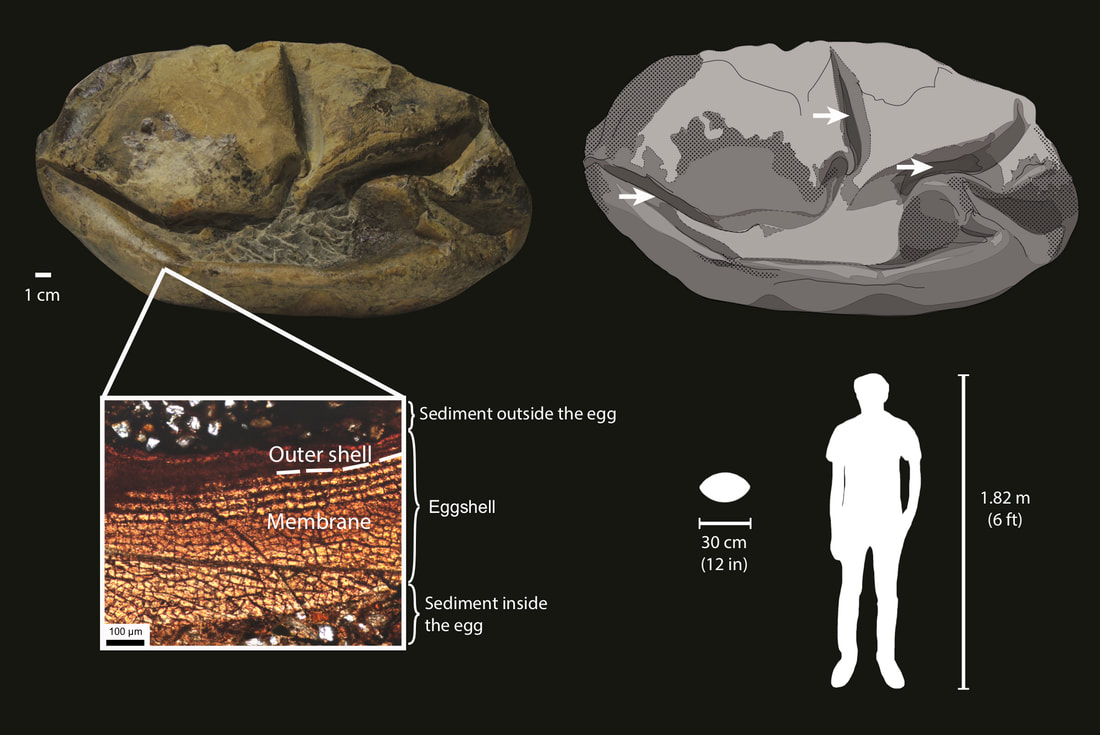
In 2020, me and my collaborators at the University of Texas at Austin and National Museum of Natural History of Chile described a giant fossilized egg from Late Cretaceous marine deposits of Antarctica (Legendre et al., 2020, Nature). The preserved eggshell shows a structure similar to those of extant lizards and snakes, with a very thin calcareous layer and a thick proteinaceous layer. This discovery was unique for two reasons. First, such an exquisitely preserved proteinaceous structure is very rare in the fossil record. Second, the eggshell is incredibly thin (~700 µm) compared to the size of the egg (~30 cm in length). This made us wonder: what animal would lay such a bizarre egg, and why?
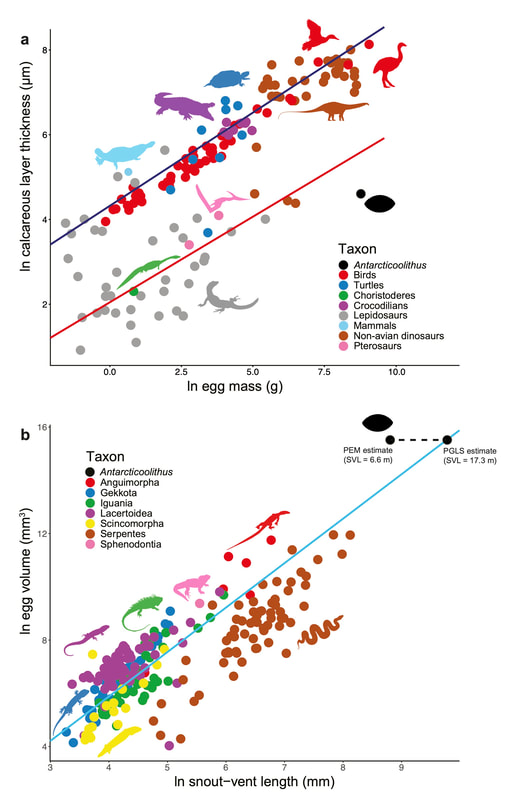
Using phylogenetic comparative methods on a dataset of various life-history traits for over 250 vertebrate species, we determined that the egg layer was most likely over 7 m in body length, compatible with the very large marine reptiles (mosasaurs and plesiosaurs) commonly found in the Late Cretaceous of Antarctica. These reptiles were viviparous, implying that the eggs hatched immediately after having been laid to allow the hatchling to go to the surface to breathe. Therefore, the eggshell likely did not have any role in maintaining the structural integrity of the egg, which would explain its highly reduced thickness and mineral content.
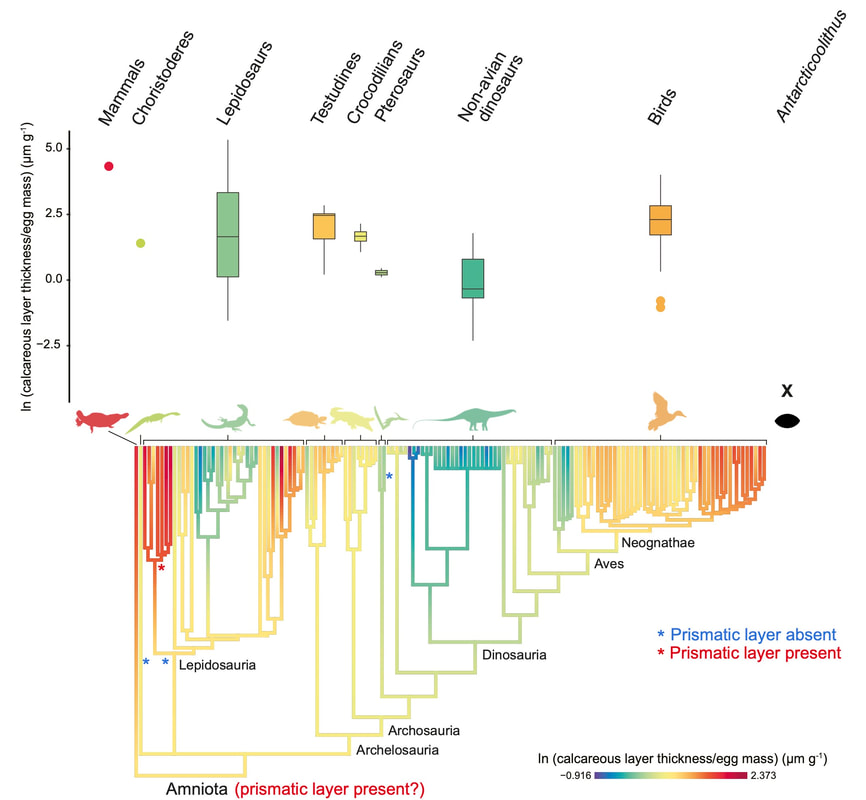
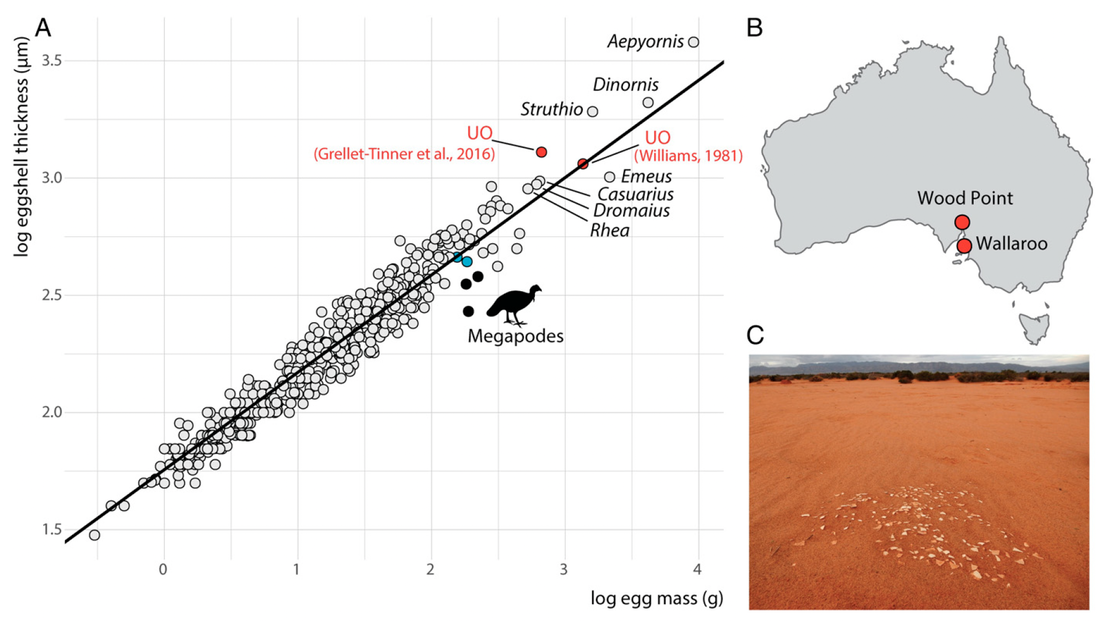
Looking at our dataset, we realized that the scaling of eggshell thickness with egg size and body size was highly variable among amniotes. We then started wondering: what could explain such a high variability? Are there any major evolutionary constraints on the evolution of eggshell thickness among reptiles?
Looking at our dataset, we realized that the scaling of eggshell thickness with egg size and body size was highly variable among amniotes. We then started wondering: what could explain such a high variability? Are there any major evolutionary constraints on the evolution of eggshell thickness among reptiles?
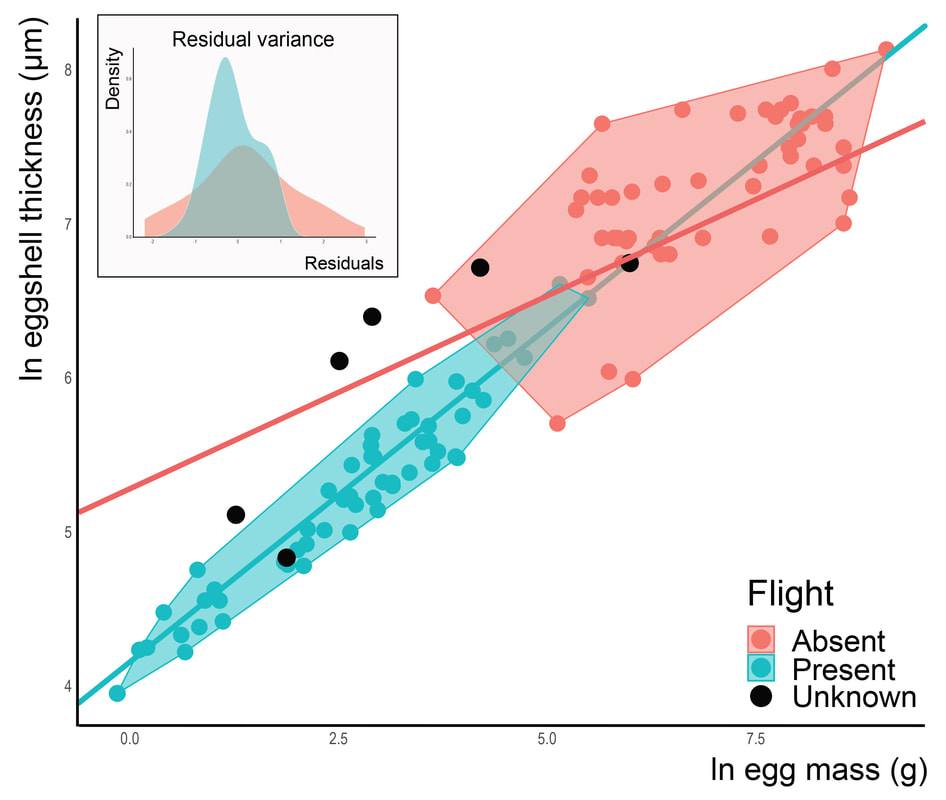
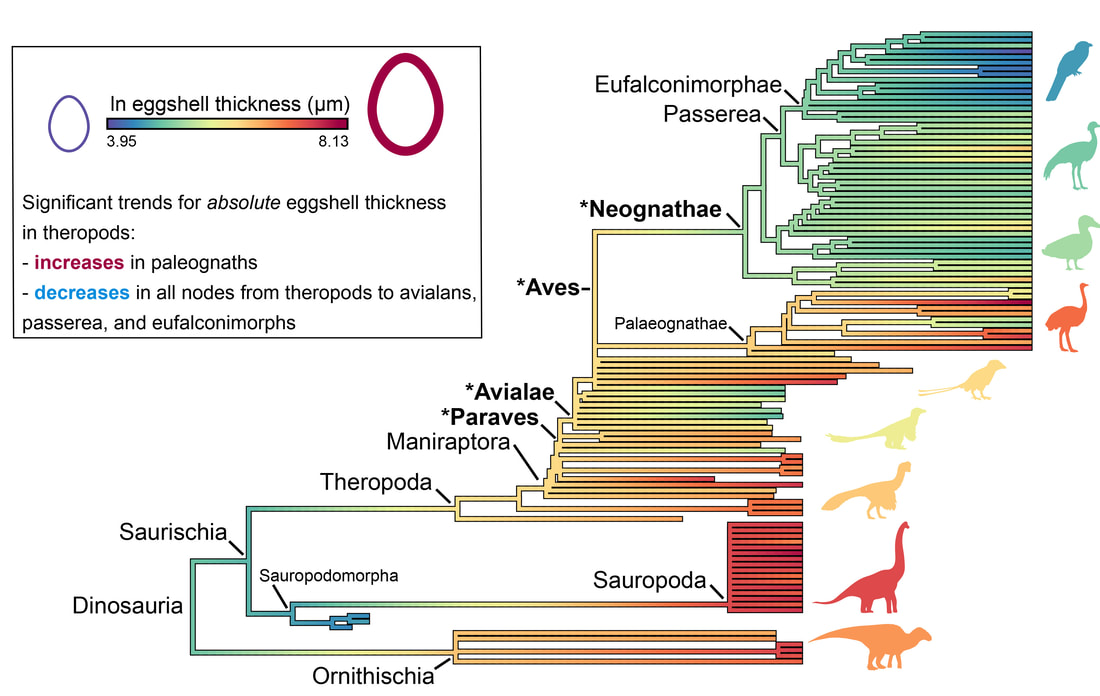
I decided to study this question in the context of dinosaur evolution, more specifically, the acquisition of the avian body plan in theropods. Using a dataset of eggshell thickness and various proposed correlates for over 100 dinosaurs, we reconstructed evolutionary rates of their eggshell thickness and identified egg mass and flight as its main correlates (Legendre & Clarke, 2021, Evolution). Indeed, the range of values for eggshell thickness in flying dinosaurs is much more constrained than that of flightless ones. We suggest that the ancestral decrease in body mass in maniraptoran dinosaurs resulted in decreased evolutionary rates of eggshell thickness in many least inclusive clades, suggesting stabilizing selection associated with the acquisition of flight among paravians. The selective pressure of high metabolic rates associated with active flight was likely a strong constraint on many life history traits, including eggshell thickness.
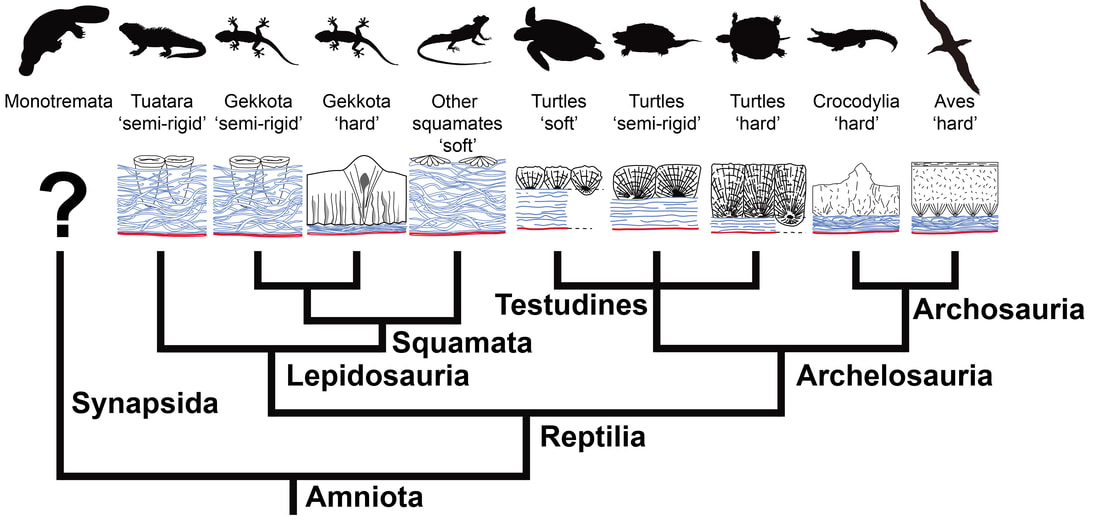
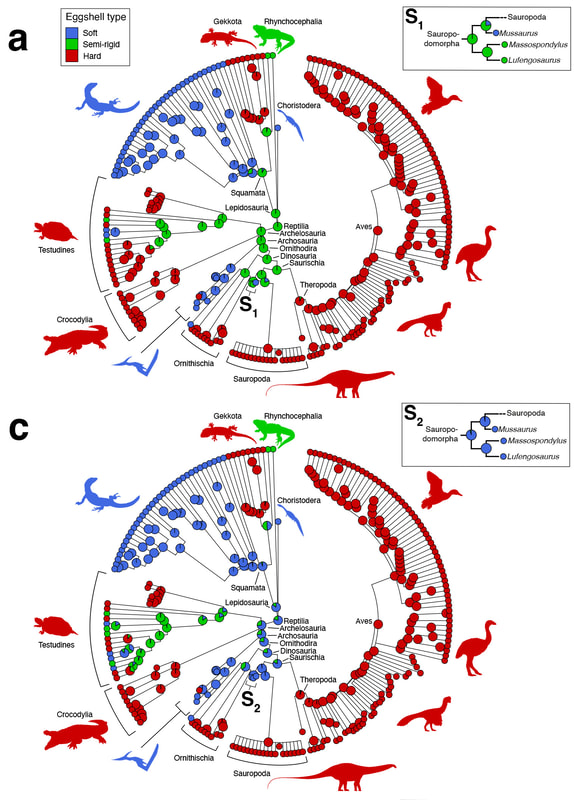
More recently, I collaborated with fossil egg expert Prof. Seung Choi (Seoul National University, South Korea) on clarifying the commonly used dichotomy of "soft-shelled" versus "hard-shelled" reptile eggs. We wrote a comprehensive review of this terminology in previous literature (Legendre et al., 2022, Journal of Anatomy), and used ancestral state reconstructions to show that this terminology is highly variable depending on authors and leads to imprecise, ambiguous results when used in a phylogenetic context. We recommended these terms to be abandoned, and defined a new set of best practices for describing eggshell microstructure in evolutionary biology.
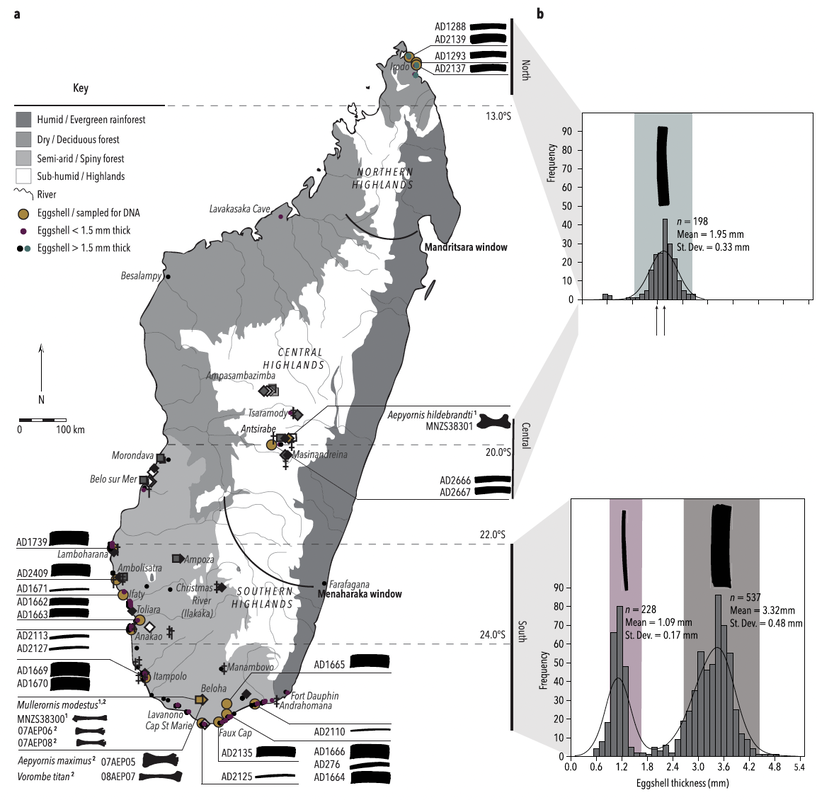
I have also collaborated on egg-related projects led by other researchers, such as combining microanatomy and paleoproteomics to attribute fossil eggshell fragments from Australia to the giant flightless bird Genyornis (Demarchi et al., 2022, Proceedings of the National Academy of Sciences), or using eggshell microstructure to understand the ecological diversification of elephant birds, the largest birds that ever lived, in Madagascar (Grealy et al., 2023, Nature Communications).
How do birds make sounds? The mysteries of the syrinx, the avian vocal organ (since 2018)
Birds have developed a unique strategy to produce sound for communication. In most terrestrial vertebrates, the organ that makes the sound is the larynx: a series of specialized cartilages, located in the throat, that support the vocal folds—thin membranes that make sound by vibrating when air is expelled from the lungs. Birds also have a larynx, but it is non-functional; instead, they make sound using a unique organ, the syrinx, which consists in a series of specialized modifications at the junction of the trachea and
bronchi—portions of the windpipe located within the ribcage. The syrinx also includes sound-producing membranes, which birds are capable of modulating the vibration of using specialized muscles. The number, position, and mechanism of action of these muscles vary a lot among different avian clades, and are not well understood in most of them, particularly in a phylogenetic context (Kingsley et al., 2018, Proceedings of the National Academy of Sciences).
bronchi—portions of the windpipe located within the ribcage. The syrinx also includes sound-producing membranes, which birds are capable of modulating the vibration of using specialized muscles. The number, position, and mechanism of action of these muscles vary a lot among different avian clades, and are not well understood in most of them, particularly in a phylogenetic context (Kingsley et al., 2018, Proceedings of the National Academy of Sciences).

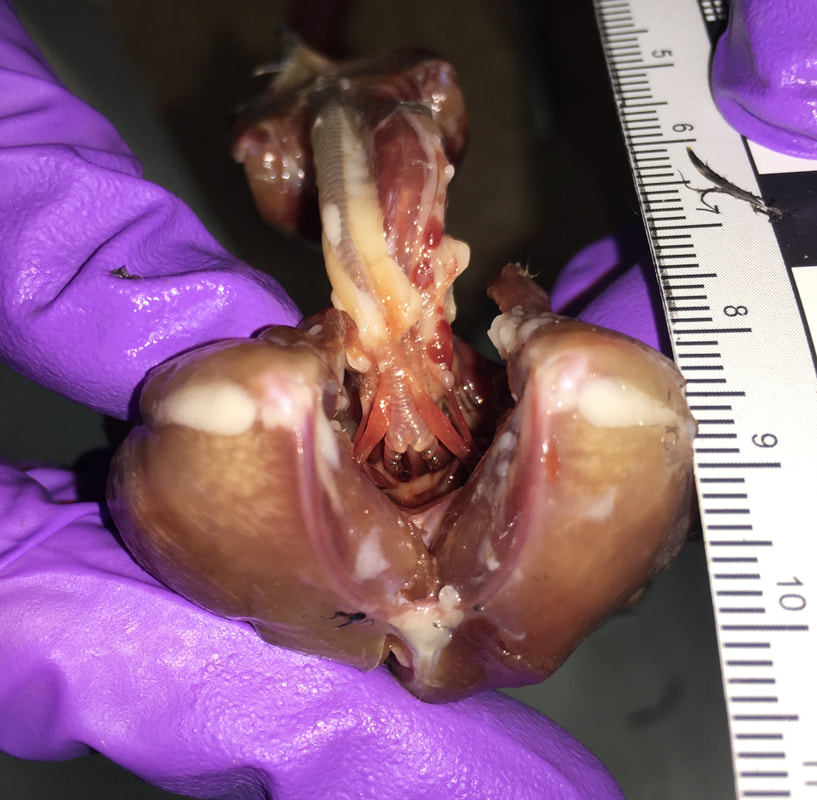
Two examples of the diversity of the avian syrinx: on the left, the syrinx of a rifleman (Acanthisitta chloris); on the right, the syrinx of a super lyrebird (Menura novaehollandiae). The syrinx consists of specialized cartilage rings in the trachea (Tr) and bronchi (Br), some of which can be fused into a tympanum (Ty). It also includes specialized muscles—including the sternotrachealis (ST) and tracheolateralis (TL), and in some species additional intrinsic muscles (IM)—that can contract to change the geometry of the vocal tract and modulate the vibration of membranes inside the syrinx to produce sound. Modified after Ames (1971, Bulletin of the Peabody Museum of Natural History).
I am a collaborator on the Syrinx Project led by Julia Clarke (The University of Texas at Austin). My research in this project has focused on the evolution of the syrinx in Strisores, a clade that includes hummingbirds, swifts, nightjars, and a number of other smaller groups (Chen & Field, 2020, Vertebrate Zoology), most of which have never been studied in the context of vocal production. Hummingbirds are one of three groups of birds—the others being songbirds and parrots— that are capable of vocal production learning (VPL) from conspecifics (ten Cate, 2021, Philosophical Transactions of the Royal Society B – Biological Sciences). VPL has been hypothesized to be linked with extended frequency range (Faiß et al., 2022, Proceedings of the Royal Society B: Biological Sciences) and highly specialized motor control of the syrinx (Riede & Olson, 2020, Scientific Reports), but its mechanisms and evolution are poorly understood in hummingbirds, especially since the syrinx of their non-learning sister group, the Apodi (swifts and treeswifts), has never been described.
In our project (Legendre et al., 2024, Zoological Journal of the Linnean Society), my collaborators and I studied the syrinx of swifts and hummingbirds using Diffusible Iodine-based Contrast-enhanced Computed Tomography (diceCT). DiceCT is the most efficient technique to produce high-contrast, high-resolution images of vertebrate soft tissues in whole-body specimens (e.g. Gignac et al., 2016, Journal of Anatomy). We provided the first general description of the swift syrinx and used phylogenetic comparative methods to identify the shortening of the trachea in hummingbirds as the likely main constraint on their acquisition of VPL. We hypothesize that shortening the trachea (and associated tracheal muscles) increased available space for intrinsic muscles on the tympanum. The associated displacement of the hummingbirds syrinx in the throat resulted in stabilization of the sound sources through stronger motor control by the surrounding intrinsic muscles, which likely facilitated high frequency vocalizing and VPL—a strategy highly distinct from that of songbirds (e.g. Monte et al., 2020, BMC Zoology).
In our project (Legendre et al., 2024, Zoological Journal of the Linnean Society), my collaborators and I studied the syrinx of swifts and hummingbirds using Diffusible Iodine-based Contrast-enhanced Computed Tomography (diceCT). DiceCT is the most efficient technique to produce high-contrast, high-resolution images of vertebrate soft tissues in whole-body specimens (e.g. Gignac et al., 2016, Journal of Anatomy). We provided the first general description of the swift syrinx and used phylogenetic comparative methods to identify the shortening of the trachea in hummingbirds as the likely main constraint on their acquisition of VPL. We hypothesize that shortening the trachea (and associated tracheal muscles) increased available space for intrinsic muscles on the tympanum. The associated displacement of the hummingbirds syrinx in the throat resulted in stabilization of the sound sources through stronger motor control by the surrounding intrinsic muscles, which likely facilitated high frequency vocalizing and VPL—a strategy highly distinct from that of songbirds (e.g. Monte et al., 2020, BMC Zoology).
The Syrinx Project has also resulted in new discoveries on the syrinx of other birds:
Additionally, I am supervising student research on syrinx anatomy and evolution - if you want to know more about current developments of the Syrinx Project, email me!
- I was a coauthor on a paper on the ostrich syrinx led by my mentee undergrad Michael Chiappone (Chiappone et al., 2024, Journal of Anatomy);
- Both Michael and Julia were coauthors on a large integrative study that identified deep homology in the developmental pathways of vocal folds in both larynx and syrinx, led by our collaborators in the Tabin Lab at Harvard University (Longtine et al., 2024, Current Biology).
Additionally, I am supervising student research on syrinx anatomy and evolution - if you want to know more about current developments of the Syrinx Project, email me!
Paleophysiological inference: reconstructing the evolution of vertebrate endothermy (2011–2014; intermittently since 2018)
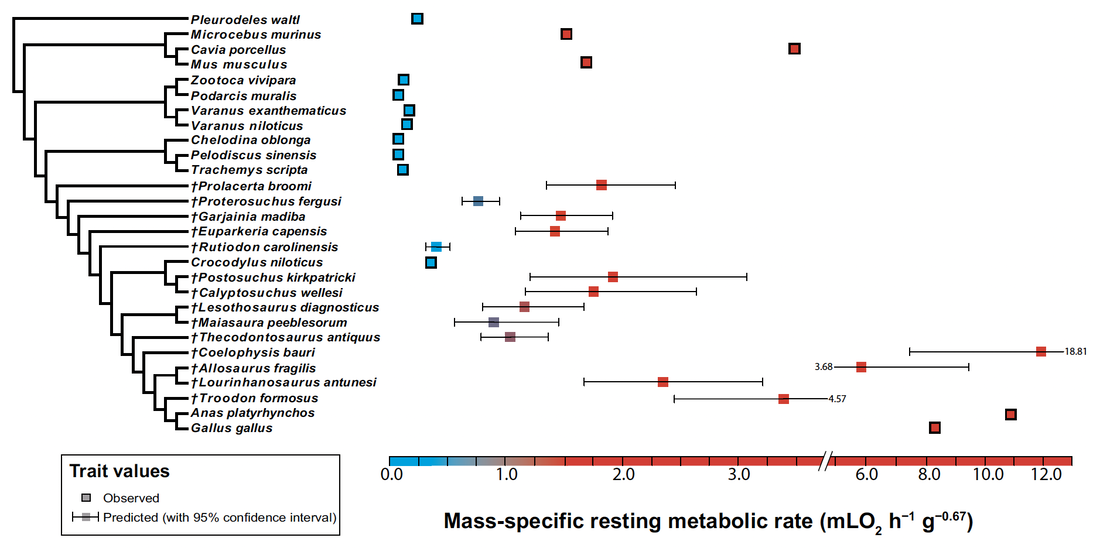
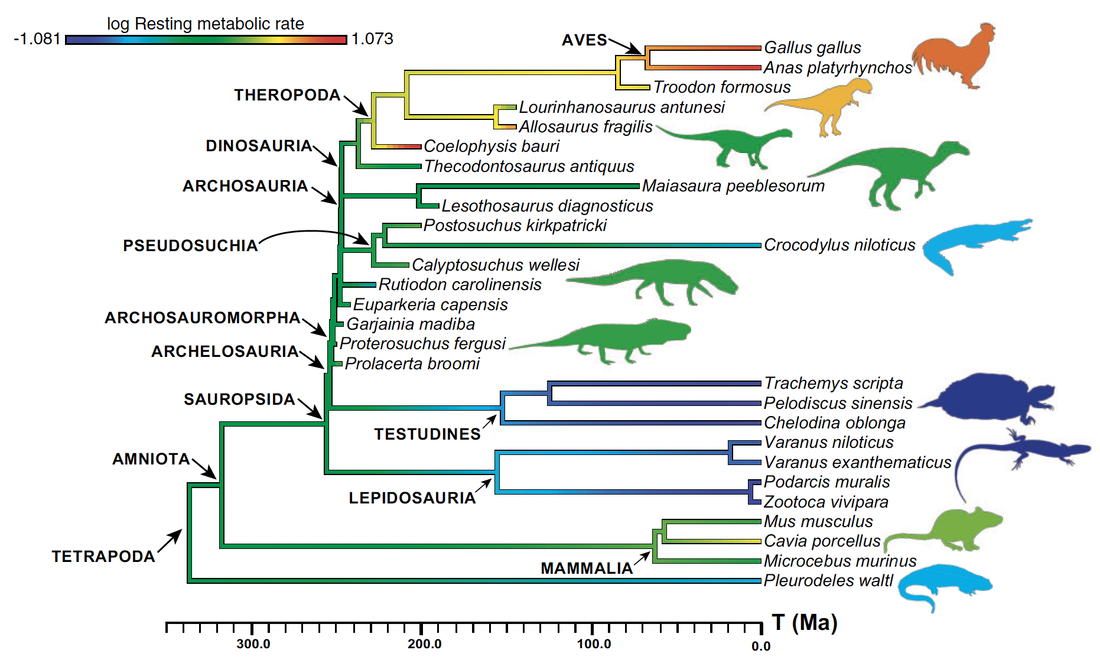
My PhD project focused on reconstructing the ancestral acquisition of endothermy (metabolic heat production) in amniotes. The ability to generate and maintain a constant body temperature, well-characterized in extant birds, had long been inferred to have arisen earlier in reptile evolution (e.g. Benton, 2021, Gondwana Research). My project, aiming at testing this hypothesis quantitatively for the first time, was highly interdisciplinary: it used histological traits measured in the limb bones of extant and extinct amniotes to build phylogenetically-informed statistical models that can predict metabolic rate in extinct archosauromorphs (e.g. non-avian dinosaurs). My initial expertise in bone microanatomy was only one in a wide array of techniques and concepts I had to learn to complete the project. Teaching myself how to use phylogenetic comparative methods in R, which no member of my lab at the time had expertise in, was especially challenging.
I collaborated with ecologist and statistician Dr. Guillaume Guénard (University of Montréal, Canada) to design a new predictive framework for metabolic rates of fossil vertebrates using phylogenetic eigenvector maps (PEM), and identified an origin of endothermy among non-archosaur archosauromorphs in the Early Triassic, i.e. over 100 million years before the earliest known birds (Legendre et al., 2016, Systematic Biology). This result is congruent with the work of other authors that used different proxies for archosaurian endothermy (e.g. Seymour et al., 2004, Physiological and Biochemical Zoology; Bourke et al., 2018, PLOS ONE; Brocklehurst et al., 2020, Philosophical Transactions of the Royal Society B; Dawson et al., 2020, Science Advances; Wiemann et al., 2022, Nature).
I collaborated with ecologist and statistician Dr. Guillaume Guénard (University of Montréal, Canada) to design a new predictive framework for metabolic rates of fossil vertebrates using phylogenetic eigenvector maps (PEM), and identified an origin of endothermy among non-archosaur archosauromorphs in the Early Triassic, i.e. over 100 million years before the earliest known birds (Legendre et al., 2016, Systematic Biology). This result is congruent with the work of other authors that used different proxies for archosaurian endothermy (e.g. Seymour et al., 2004, Physiological and Biochemical Zoology; Bourke et al., 2018, PLOS ONE; Brocklehurst et al., 2020, Philosophical Transactions of the Royal Society B; Dawson et al., 2020, Science Advances; Wiemann et al., 2022, Nature).
Later studies by other authors have since used my PEM framework to infer the paleophysiology of other vertebrate clades, such as synapsids (Olivier et al., 2017, Biological Journal of the Linnean Society; Faure-Brac & Cubo, 2020, Philosophical Transactions of the Royal Society B), plesiosaurs (Fleischle et al., 2018, PeerJ), and pseudosuchians (Cubo et al., 2020, Biological Journal of the Linnean Society; Faure-Brac et al., 2022, Paleobiology).
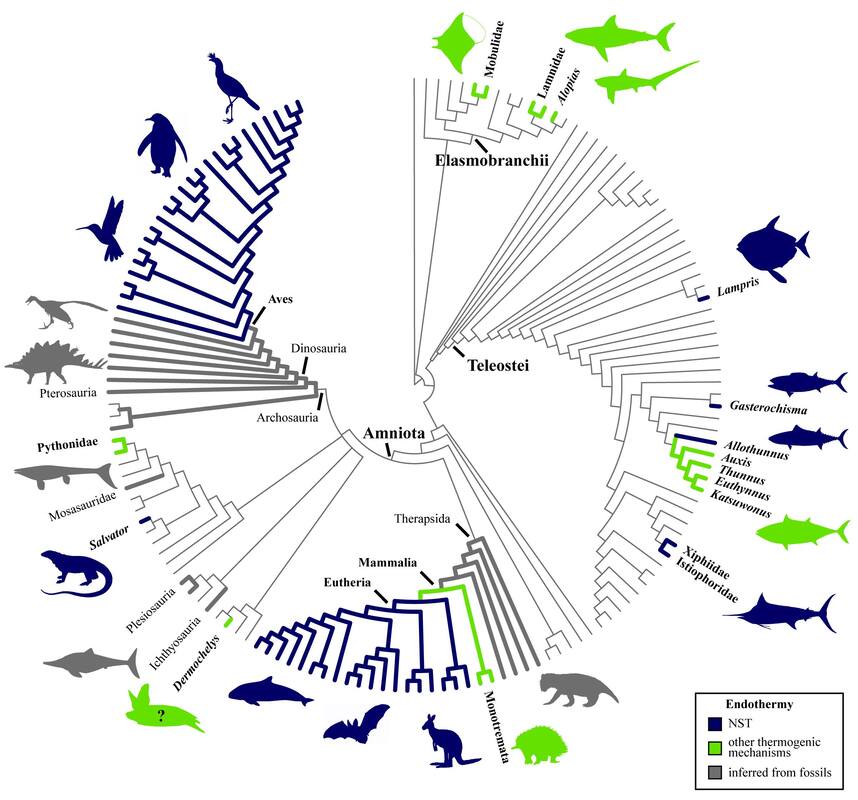
In 2020, I collaborated with ichthyologist Dr. Donald Davesne (Museum für Naturkunde, Berlin, Germany), on a detailed review of the evolution of vertebrate endothermy (Legendre & Davesne, 2020, Philosophical Transactions of the Royal Society B), which was the first phylogenetically-informed review of such physiological adaptations for all vertebrates. I still collaborate with paleontologists and physiologists on projects linked with vertebrate endothermy, and review papers in the field.
In 2020, I collaborated with ichthyologist Dr. Donald Davesne (Museum für Naturkunde, Berlin, Germany), on a detailed review of the evolution of vertebrate endothermy (Legendre & Davesne, 2020, Philosophical Transactions of the Royal Society B), which was the first phylogenetically-informed review of such physiological adaptations for all vertebrates. I still collaborate with paleontologists and physiologists on projects linked with vertebrate endothermy, and review papers in the field.
Influence of fossoriality on bone microstructure in mammals: a case study on the aardvark (2016–2018)
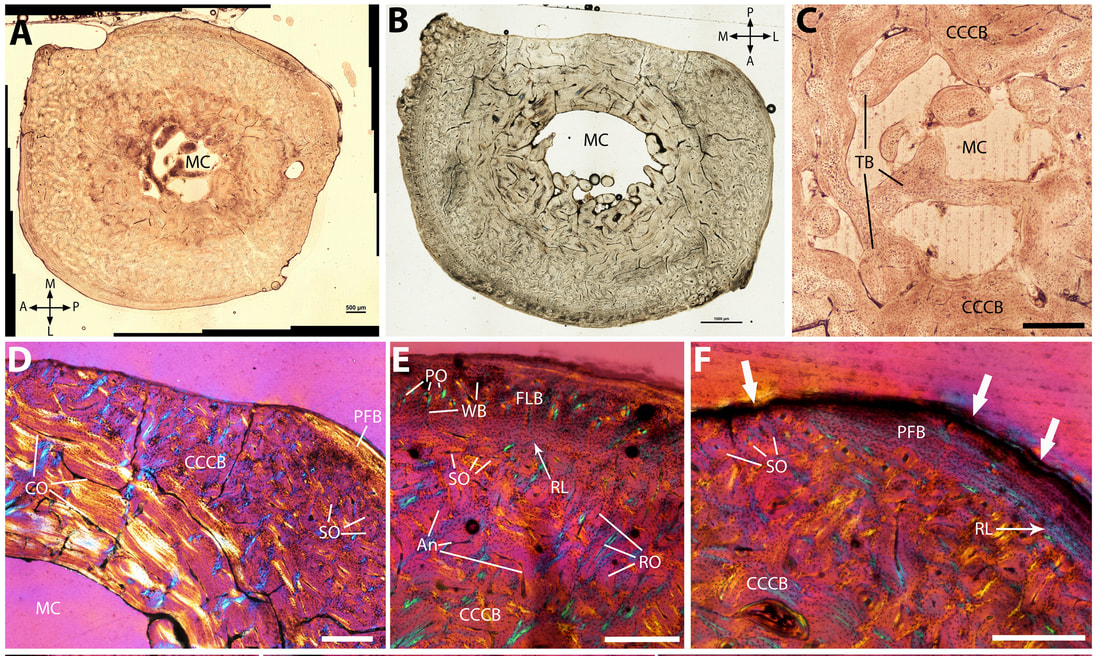
I did a two-year postdoctoral research project at the National Museum, Bloemfontein, South Africa, working with Dr. Jennifer Botha. We studied the limb bone microstructure of the aardvark (Orycteropus afer), the largest extant fossorial mammal, and discovered that the cortex of all its limb bones was mostly composed of compacted coarse cancellous bone (CCCB), a poorly known bone tissue type that tends to get completely resorbed throughout ontogeny in vertebrates due to its low biomechanical resistance (Legendre & Botha-Brink, 2018, PeerJ). This result was totally unexpected: since aardvarks are highly specialized scratch diggers that burrow almost every day for food and shelter, their limbs are required to support strong biomechanical constraints associated with digging behavior. We proposed a new hypothesis to explain this paradox: the low metabolic rate of aardvarks, in part explained by their fossorial lifestyle in a cold, underground environment and by a diet with low nutritive value (ants and termites), prevents them from replacing CCCB with dense haversian bone throughout ontogeny. To compensate, aardvarks have developed strong cortical drift in their limb bones and very limited pronation and supination of the forelimb to limit torsional and bending constraints.
Based on these results, we suggested that the presence of CCCB in mammals might be an adaptation to fossoriality. This hypothesis has since been confirmed by other authors who identified high amounts of CCCB in other fossorial mammal groups, such as armadillos (Heck et al., 2019, PLOS ONE), wombats (Walker et al., 2021, Australian Mammalogy), and true moles (Amson et al., 2022, Evolution Letters).
Based on these results, we suggested that the presence of CCCB in mammals might be an adaptation to fossoriality. This hypothesis has since been confirmed by other authors who identified high amounts of CCCB in other fossorial mammal groups, such as armadillos (Heck et al., 2019, PLOS ONE), wombats (Walker et al., 2021, Australian Mammalogy), and true moles (Amson et al., 2022, Evolution Letters).
|
|
Dr. Lucas Legendre
Research Associate Department of Earth and Planetary Sciences The University of Texas at Austin 2305 Speedway Stop C1160 Austin, TX 78712, United States [email protected] All pictures on this website belong to me, unless mentioned otherwise. |